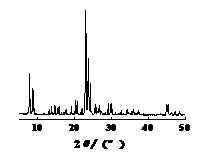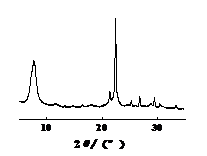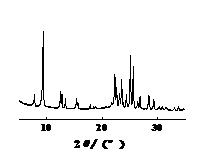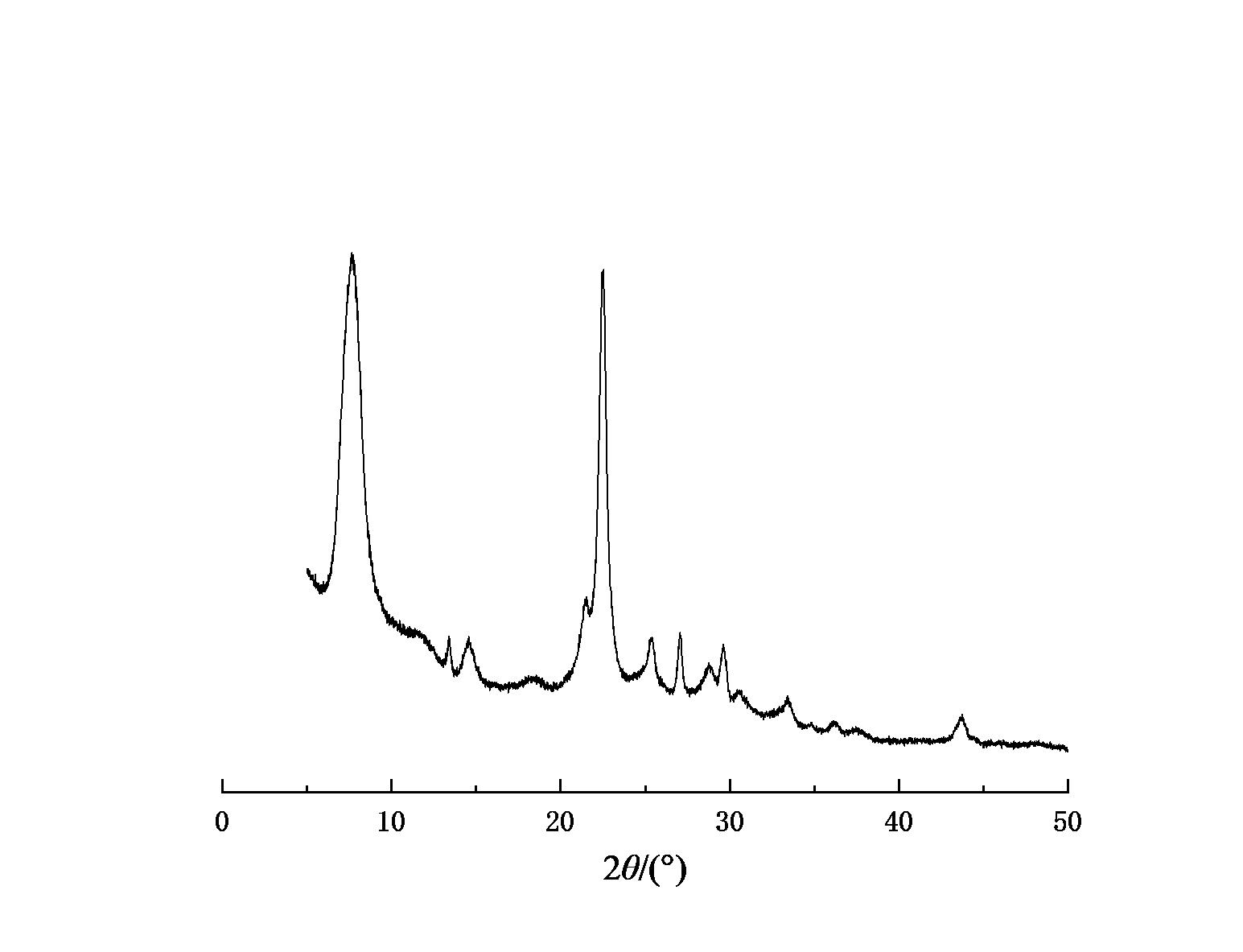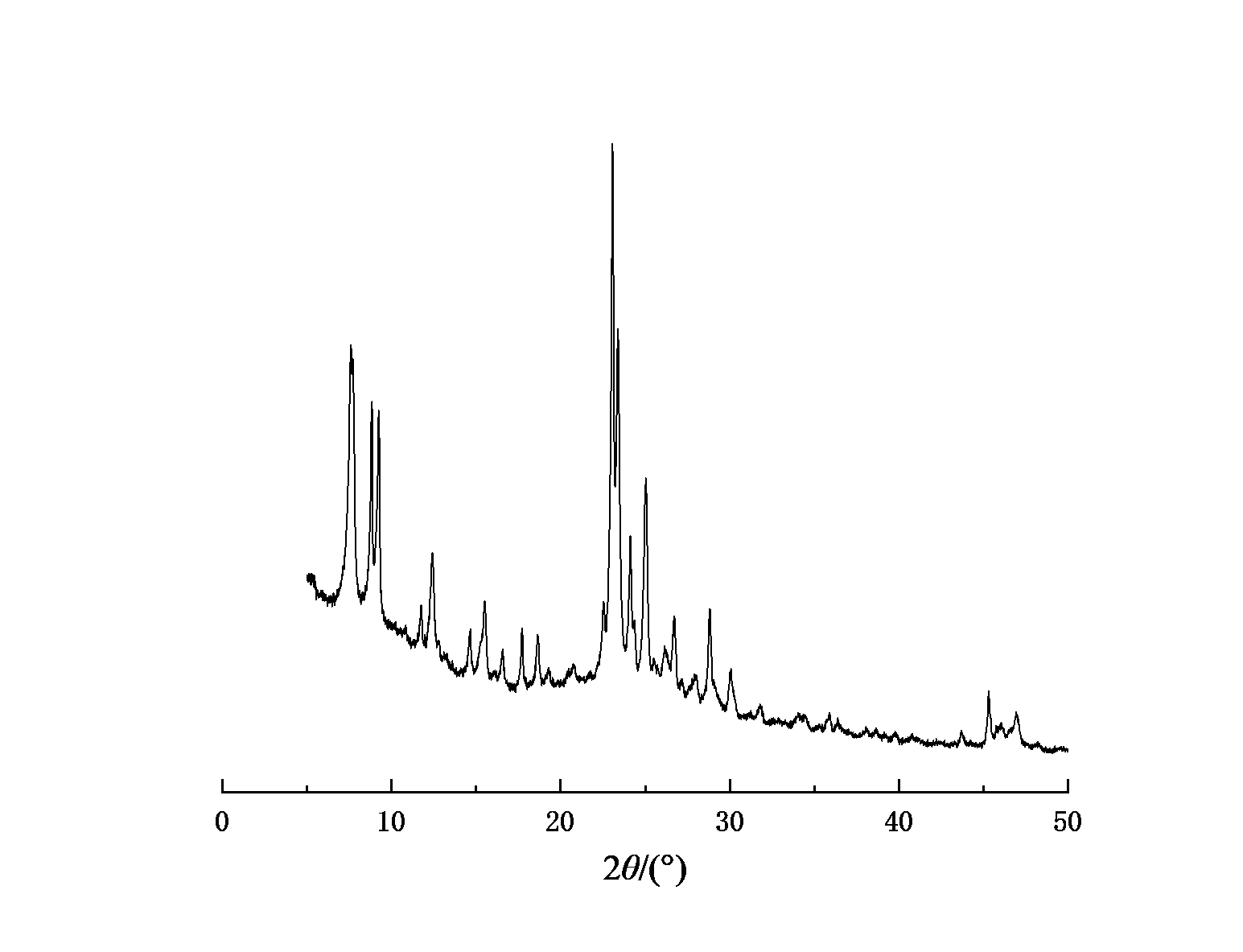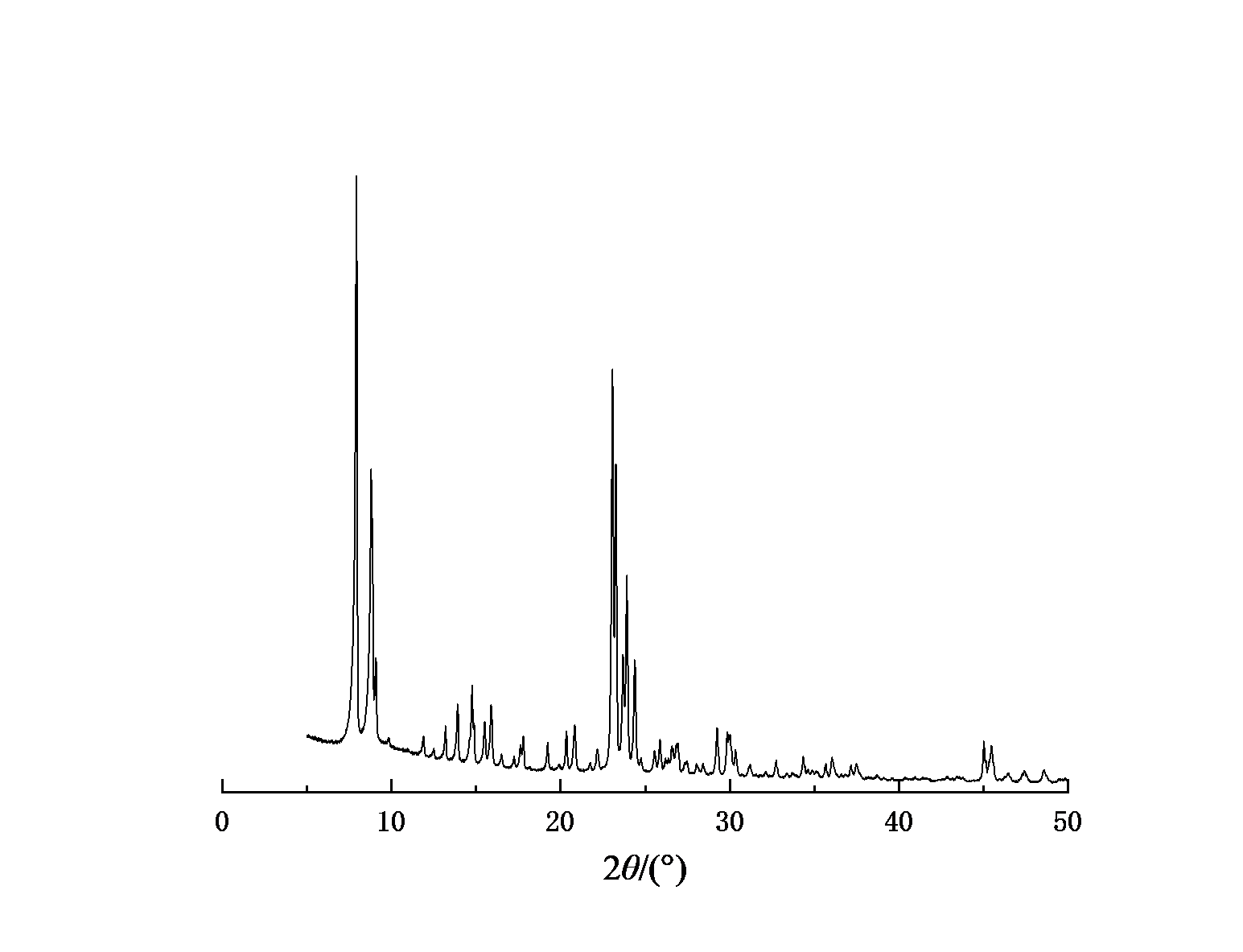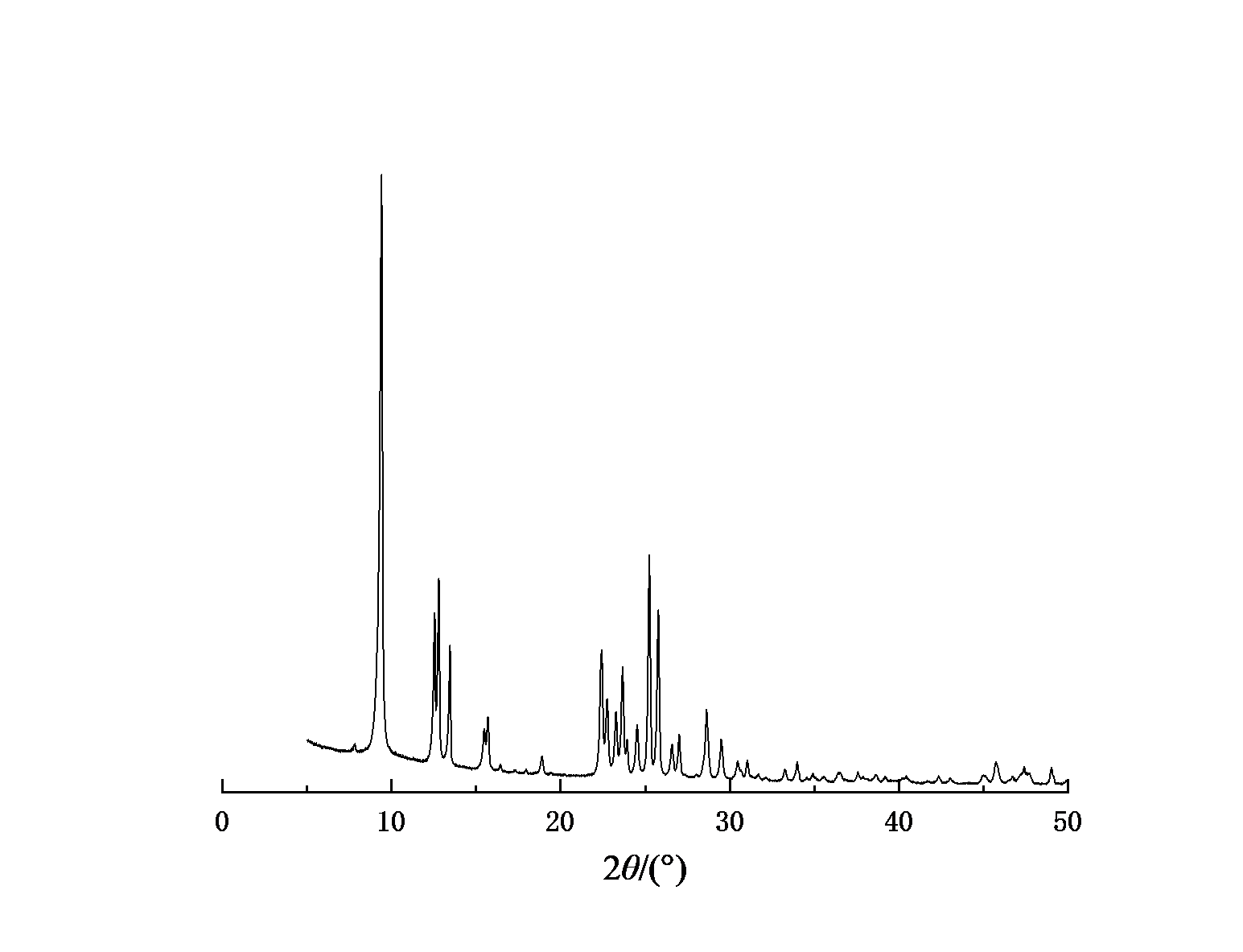1. Introduction
Chemical Oxygen Demand (COD) is a chemical method used to measure the amount of reducible substances that need to be oxidized in water samples. It is an important indicator for evaluating the degree of organic pollution in water bodies and is also one of the eight wastewater pollutant indicators for implementing total discharge control. In every stage of wastewater treatment, the frequency of COD monitoring is very high. In the detection process of CODCr in the national standard GB11914-89, oxidants such as potassium dichromate, catalysts such as silver sulfate, and masking agents such as mercury sulfate need to be added. There are some toxic chemicals such as soluble silver salts and mercury salts in the measured wate liquid. Heavy metal ions have characteristics such as high toxicity, enrichment, and persistence[1-3]. According to the GB11914-1989 CODCr monitoring method, the preparation of sulfuric acid silver sulfate reagent required for CODCr determination of wastewater chemical oxygen demand is to add 5g of silver sulfate to 500mL of concentrated sulfuric acid, which means that 100mL of this reagent contains 1g of silver sulfate. For each 150mL waste water sample, 30mL of sulfuric acid silver sulfate reagent, i.e. 0.3g of silver sulfate, with a theoretical content of 0.208g of Ag and a silver ion concentration of 1-1.5g/L, needs to be added. If discharged directly without treatment, it not only pollutes the environment but also causes waste of metal resources such as silver. Because zeolite has a large porosity and a large specific surface area, it is a substance with good adsorption performance, which can effectively adsorb heavy metal ions in aqueous solution and can be used for water purification, zeolite adsorption has the advantages of selective adsorption and high-efficiency adsorption[4-9]. Therefore, exploring efficient methods for treating heavy metal ions in COD monitoring wastewater has significant economic and environmental implications.
2. Technical processes and experiments
Select ZSM-5 molecular sieve, Beta molecular sieve, IM-5 molecular sieve, FER molecular sieve, etc. as adsorbents, simulate water samples of heavy metal ions, and explore the adsorption relationship between molecular sieves and heavy metal silver ions and mercury ions. The structure of the molecular sieve was determined by XRD diffraction before and after adsorption of heavy metal ions and the changes in the content of heavy metal ion over the molecular sieves before and after adsorption were analyzed by XRD diffraction. X-ray fluorescence spectroscopy was used to analyze the changes of metal ion content before and after molecular sieve. The content of Ag in the filtrate was analyzed by ICP; TEM was used to characterize the distribution of Ag in zeolite.
3. Results and Discussion
3.1. Characterization of Molecular Sieve Structure before adsorption
3.1.1. XRD diffraction analysis of molecular sieves
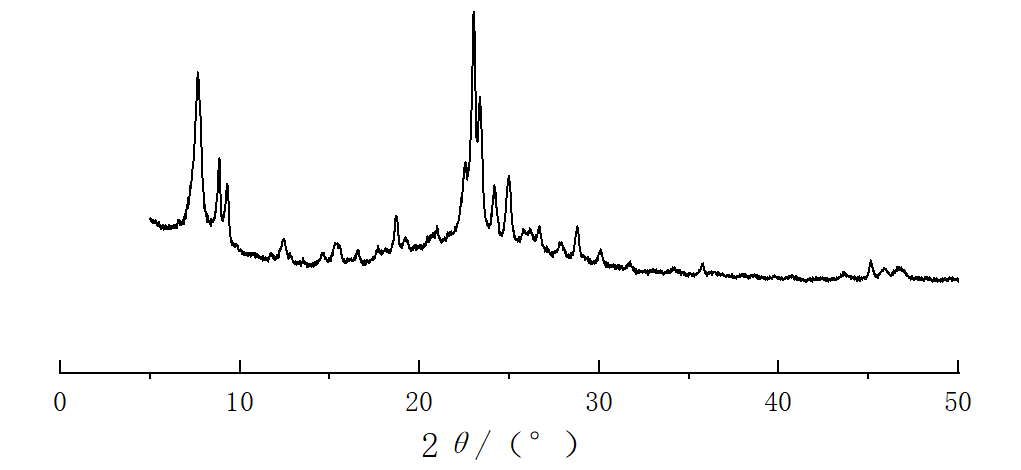
Figure 1-4 shows the XRD spectra of four types of molecular sieves, Beta, ZSM-5, IM-5, and ZSM-35. It can be seen from the figures that the crystals of the four types of molecular sieves are regular and have high crystallinities.
|
|
Figure 1: XRD spectrum of IM-5 molecular sieve | Figure 2: XRD spectrum of ZSM-5 molecular sieve |
|
|
Figure 3: XRD spectrum of Beta molecular sieve | Figure 4: XRD spectrum of molecular sieve |
3.1.2. Specific surface area, pore volume distribution and XRF analysis
Table 1 shows the specific surface area and pore volume of Beta, ZSM-5, IM-5, and ZSM-35 molecular sieves. From the table1, it can be seen that Beta has a high specific surface area and pore volume, while there is hardly any difference among the other three types of molecular sieves.
Table 1: Specific surface area and pore volume of different molecular sieves
sample | n(SiO2)/n(Al2O3) | Stotal/ m2•g-1 | Smicro/ m2•g-1 | Smeso/ m2•g-1 | Vtotal/ cm3•g-1 | Vmicro/ cm3•g-1 | Vmeso/ cm3•g-1 |
IM-5 | 36 | 337 | 287 | 50 | 0.345 | 0.359 | 0.131 |
ZSM-5 | 47 | 379 | 368 | 11 | 0.191 | 0.171 | 0.020 |
Beta | 23 | 599 | 507 | 92 | 0.460 | 0.230 | 0.230 |
FER | 70 | 308 | 215 | 93 | 0.405 | 0.097 | 0.308 |
Table 2 shows the elemental analysis of Beta, ZSM-5, IM-5, and ZSM-35 molecular sieves, all of which mainly contain silicon and aluminum elements, with low or undetectable content of other metal elements.
Table 2: Elemental analysis of different molecular sieves
sample | Na2O | Al2O3 | SiO2 | n(SiO2)/n(Al2O3) | Ag2O |
ZSM-5 | 0.0154 | 2.440 | 97.400 | 68 | --- |
ZSM-35 | 00320 | 7.720 | 92.000 | 20 | --- |
IM-5 | 0.0292 | 3.310 | 96.300 | 49 | --- |
Beta | 0.000 | 4.550 | 95.300 | 36 | --- |
3.2. Simulate the treatment of COD detection waste liquid
According to GB11914-1989 CODCr monitoring method, the preparation of sulfuric acid silver sulfate reagent required for CODCr determination of wastewater chemical oxygen demand is to add 5g of silver sulfate to 500mL of concentrated sulfuric acid, which means that 100mL of this reagent contains 1g of silver sulfate. According to statistics, for every 150mL waste water sample measured, 30mL of sulfuric acid silver sulfate reagent is required, which is 0.3g of silver sulfate with a theoretical content of 0.208g of Ag and a silver ion concentration of 1-1.5g/L.
The solubility of silver sulfate is relatively low, with only 0.836g dissolved per 100g of water at 25 ℃. According to the approximate concentration of silver ions in COD waste liquid, prepare 1L of 1.5g/L silver ion standard solution as the research object. Four types of molecular sieves were weighed with different masses to study the effect of different molecular sieves on the treatment of silver ions in simulated waste liquid.
3.3. Characterization of Molecular Sieve Structure after adsorption
3.3.1. XRD analysis
|
|
Figure 5: XRD spectrum of Beta molecular sieve | Figure 6: XRD spectrum of IM-5 molecular sieve |
|
|
Figure 7: XRD spectrum of zsm-5 molecular sieve | Figure 8: XRD spectrum of ZSM-35 molecular sieve |
From the XRD spectra of the four types of molecular sieves shown in Figures 5-8, it can be seen that the crystal structures of the four types of molecular sieves remain intact after treating simulated wastewater. This indicates that silver ions have not destroyed the essential structure of the molecular sieves, which is beneficial for the reuse of the molecular sieves.
3.3.2. XRF analysis
Table 3: XRF analysis of four types of molecular sieves
sample | Al2O3(ω%) | SiO2(ω%) | Na2O(ω%) | Ag2O(ω%) |
Beta | 9.35 | 90.50 | 0.0208 | 1.85 |
ZSM-5 | 3.50 | 94.10 | 0.0354 | 1.75 |
IM-5 | 4.57 | 92.50 | 0.0397 | 2.55 |
ZSM-35 | 2.46 | 95.30 | 0.0219 | 1.89 |
Table 3 shows the elemental analysis of simulated wastewater treated with Beta, ZSM-5, IM-5, and ZSM-35 molecular sieves. From Table 3, it can be seen that the four types of molecular sieves mainly contain silicon and aluminum elements. Before and after treating simulated wastewater, there is a significant change in the content of silver element, among the molecular sieves, while the IM-5 molecular sieve adsorbs the highest silver ion content, reaching 2.55%, whose removal rate can reach over 60% after calculation. Molecular sieves are the active centers of catalysts, and the adsorbed silver ions can be used as raw materials for the synthesis of highly active molecular sieves containing heavy metals.
3.3.3. TEM analysis




Figure 9: TEM analysis of IM-5 molecular sieve
From Figure 9, it can be seen that the crystal structure of IM-5 molecular sieve is regular, with a large number of aggregated particles observed macroscopically. The pore size of IM-5 molecular sieve is limited. From the comparison of the scale, larger aggregated particles should be adsorbed on the surface of IM-5 molecular sieve, and there are fewer aggregated particles entering the interior of the molecular sieve. And these aggregated particles are mainly composed of silver compounds. This provides the possibility of reusing molecular sieves and recovering silver ions at high concentrations.
3.3.4. ICP analysis
Table 4: ICP-AES analysis of simulated wastewater samples treated with four types of molecular sieves
Na(mg/kg) | Al(mg/kg) | Si(mg/kg) | Ag(mg/kg) | |
Beta | 1.40 | 11.00 | 4.90 | 635 |
ZSM-5 | <1.00 | <1.00 | <1.00 | 662 |
IM-5 | 4.90 | <1.00 | <1.00 | 570 |
ZSM-35 | 1.20 | 2.2 | <1.00 | 600 |
Table 4 shows the elemental analysis of simulated wastewater samples treated with Beta, ZSM-5, IM-5, and ZSM-35 molecular sieves. From Table 4, it can be seen that the content of silver ions in the water sample has significantly decreased, with IM-5 molecular sieve showing the largest decrease, and the other three molecular sieves also showing a decrease of at least 50% or more. This is consistent with the elemental XRF analysis results of the four molecular sieves.
4. Conclusion
Through TEM analysis of IM-5 molecular sieve used to treat COD waste liquid, a large amount of silver ions accumulated on the surface of the molecular sieve and did not enter the pore channels of the molecular sieve. This indicates that silver ions are more easily adsorbed on the surface of the molecular sieve. Silver ions have a larger water and radius, and their movement within the pore channels of the molecular sieve is greatly restricted compared to the outer surface. This provides the possibility of reusing molecular sieves and recovering high concentrations of heavy metal ions.