1. Introduction
With the development of global industry, an increasing amount of greenhouse gas is emitted into the atmosphere. Though there are already many kinds of renewable energy sources, fossil fuel still plays an essential role in worldwide energy consumption, which results in a more intense greenhouse effect. Therefore, it’s of great importance to create reusable and efficient energy storage devices to cut down on the global carbon footprint, as renewable and alternative energy sources are essential to solving twin problems, energy and climate change [1-3]. Supercapacitor is an eco-friendly energy storage system, gaining significant attention in the field of high-performance energy storage [4]. As the research into carbon material has received great success, there is much space for supercapacitors to develop and satisfy the need for energy by storing energy in a more efficient and green way [5].
Batteries and supercapacitors are two major methods of energy storage. The batteries transfer chemical energy into electricity while the capacitors store energy mainly via the non-Faraday process. By adding voltage to two large-surface metal sheets separated by insulators, supercapacitors store energy and have a much higher lifespan than batteries due to their highly reversible process. However, supercapacitors cannot provide a stable voltage while batteries can. The energy density of supercapacitors remains significantly lower compared to batteries, with electrochemical capacitors achieving less than 10% of the energy density of advanced batteries [6]. Compared with conventional capacitors, Supercapacitors achieve higher energy densities while preserving their distinctive high power density [7].
An asymmetric capacitor typically uses a set of electrode materials with great specific surface area and another set with a high Faradaic capacitance effect [8]. Apart from EDLCs which completely store energy by non-Faraday process, asymmetric capacitors not only employ the electric double-layer effect for energy storage but also adopt energy storage through electrochemical reactions, resulting in improved storage performance demonstrated by proper pairing and balancing two asymmetric electrodes [9]. Batteries rely on redox reactions to store energy, resulting in higher energy density. In contrast, supercapacitors rely on the electric double-layer effect, which leads to lower energy density but higher power density. By combining these two mechanisms, asymmetric capacitors significantly improve energy density compared to symmetric capacitors. At the same time, since the two electrode materials of an asymmetric capacitor have different electrochemical potential windows, the assembled asymmetric capacitor can have a wider overall voltage range [10]. The design of asymmetric capacitors can significantly expand the operating voltage range of the capacitor.
The research designed a solid-state supercapacitor. MXene, known as transition metal carbides and nitrides [11], is combined with the CPVP-A composite as the anode. As one of the most prospective 2-dimensional materials, MXene offers benefits such as a rich surface, functional groups, metallic conductivity, high specific surface area, and excellent hydrophilicity[12,13]. Compared with traditional methods to synthesize Mxene such as polymer carbonization, alkali treatment, and template-sacrificing method, preparing Mxene in this research improves the three mentioned methods to increase Capacitance, rate performance, and cycle stability by lowering the temperature of preparation, adding no additional agents and improving the alkali treatment [14].
The cathode of the solid-state capacitor is composed of MnO2/PEDOT: PSS nanostructured composite. As MnO2 has a high theoretical specific capacitance and is abundant in nature [15], it can be utilized in the solid-state capacitor. However, the biggest disadvantage of MnO2 is its poor electroconductivity. PEDOT:PSS, with excellent cheapness and commonality, has a high electrical conductivity but its electrochemical activity is not strong enough to meet the supercapacitor requirements [16]. Therefore, by mixing PEDOT:PSS with metal oxides, namely MnO2, the electrode is effective when Functioning as both a conductive additive and a polymer adhesive in the supercapacitor [17,18]. The electrolyte of the solid-state supercapacitor is made of chitosan and polyethylene oxide. As one of the most abundant natural polymers, chitosan costs less than human-synthesized polymer materials [19]. Moreover, chitosan has a good affinity for transition metal ions and can be molded into different shapes for various utilization. In this research, 70 wt% chitosan-30 wt% PEO is combined with lithium perchlorate (LiClO4) [20].
2. Experiments
2.1. Preparation of cathode
To fabricate the MnO2/PEDOT composite, a stainless-steel wire grid undergoes a special preparation process. Initially, the wire mesh is thoroughly cleaned via ultrasonic treatment in a 50-50 blend of acetone and ethanol to ensure a surface with no contamination. Subsequently, rectangular electrodes of 1.5 × 2.0 cm² and approximately 64 cm substantial thickness are employed. The co-electrodeposition step is essential in the fabrication, involving an aqueous solution containing 80 mL of 0.1 M manganese (II) acetate and 0.4 mL of a 1 wt% PEDOT solution. The process happens under the condition of 10.0 V DC voltage. Once the electrode is successfully formed, it goes through a rinsing process with deionized water to eliminate any residuals. Finally, the electrode is dried at a moderate temperature of 60°C for one hour to complete the MnO2/PEDOT composite [18].
2.2. Preparation of anode
To synthesize Ti3C2Tx MXene, a blend of HCl and LiF was used along with 1 g Ti3AlC2. The mixture, of 9 M HCl and 1.98 g LiF was agitated at 40 °C for 45 hours. After this step, multiple centrifugation cycles at 3500 rpm (each lasting 5 minutes) were performed until the pH surpassed 6, producing a rich solution containing multilayer Ti3C2Tx MXene [12].
After the final centrifugation, the collected sediment was dispersed in 300 mL of deionized water. The solution was then deoxygenated with an Ar flow for 30 minutes and sonicated in an ice bath for an hour at room temperature with an Ar atmosphere. The final centrifugation step lasted one hour at 3500 rpm. The result was a dark green gel suspension comprising layered Ti3C2Tx MXene flakes, poised for the preparation of diverse Ti3C2Tx MXene-based electrodes.
To fabricate the electrode material, a Ti3C2Tx MXene suspension was thoroughly blended with anhydrous ethanol, maintaining the volume ratio of 3:1. This mixture was stirred for ten minutes. After finishing this step, PMMA nanospheres were added into the solution, followed by an additional half-hour of stirring. A PVP solution with a 2 wt% concentration was combined with a solid mass ratio of 0.3:1 relative and added to the existing mixture. Using a polypropylene membrane, specifically 3501 coated PP from Cel-gard, the mixture was filtered to yield a uniform film. This film underwent a soaking treatment in a KOH solution for two hours. Afterward, it was gently dried at room temperature and separated from its supportive membrane. The composite film was exposed to intense heat at 280 °C under the flow of Ar for two hours to stabilize its structure. Subsequently, annealing was conducted at 400 °C under Ar flow for another four hours, resulting in an electrode with a thickness ranging between 3 and 6 micrometers, exhibiting a refined structure and enhanced properties suitable for advanced applications.
2.3. Preparation of the chitosan-polyethylene oxide electrolyte
The materials were from Sigma-Aldrich, including high molecular weight chitosan (CS) with an average molecular weight ranging from 310,000 to 375,000, and polyethylene oxide (PEO) powder of 300,000 molecular weight in average. To produce a polymer blend of CS and PEO in a 70:30 ratio, both polymers were individually dissolved in 50 mL of 1% acetic acid solution at room temperature for two hours. Then, the CS-PEO solutions were merged and thoroughly stirred for three hours to blend thoroughly. Once the blending process was completed, LiClO4 was added into the mixture in increments of 10%, up to a total concentration of 40% with continuous stirring. After this step, the mixture was poured into labeled Petri dishes and dried naturally at room temperature. For additional thorough drying, the films were then placed in a desiccator to eliminate any traces of moisture. This approach ensured that the films were entirely solvent-free, utilizing the blended polymers as the primary structural materials.[20].
2.4. The Fabrication and test method of Asymmetric Aqueous Supercapacitor
The anode of the asymmetric supercapacitor is MXene/CPVP-A composite electrode, while the cathode is made of MnO2/PEDOT: PSS Nanostructured Composite. The two electrodes are packed with 40 wt% as electrolyte and a polymer (CS-PEO 25Cm) in between. The mass loading of MnO2/PEDOT: PSS and activated carbon are 0.94 mg/cm2 and 0.99 mg/cm2, respectively. PET films and EVA hot melt glue packaged the supercapacitor [18].
The raw data of the test result is generated by the EClab 10.4 software platform and then converted into processable data formats. The figures below are created on the basis of these data formats using Origin 2024 as the drawing tool.
3. Results & Discussion
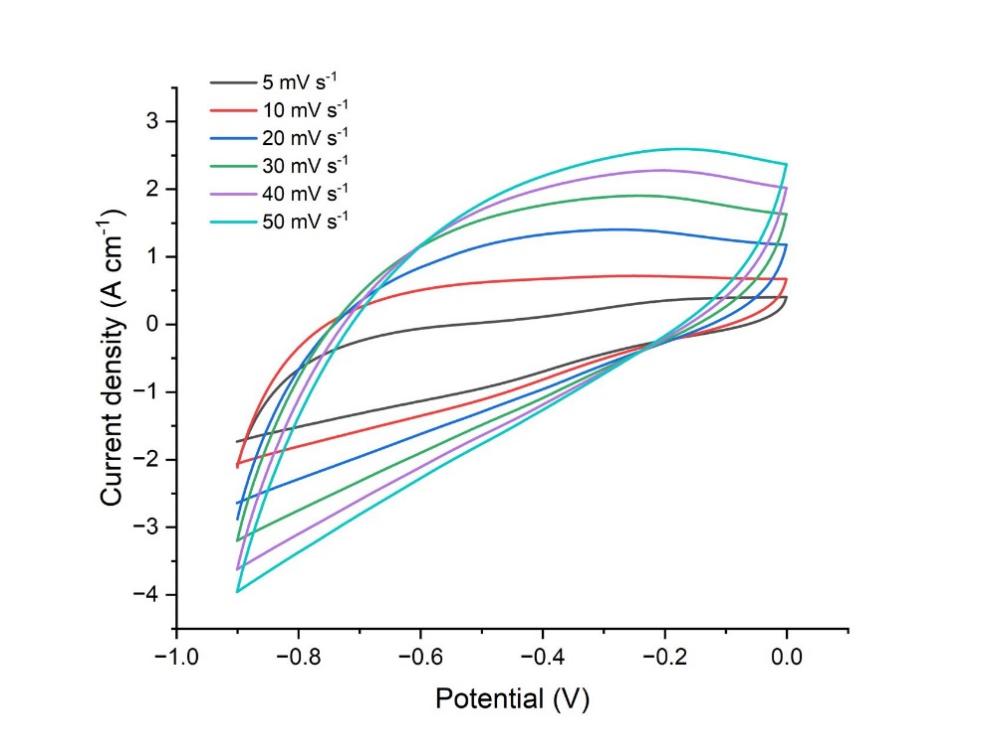
Figure 1: CV curves of anode at different scan rates ranging from 5 mV s-1 to 50 mV s-1.
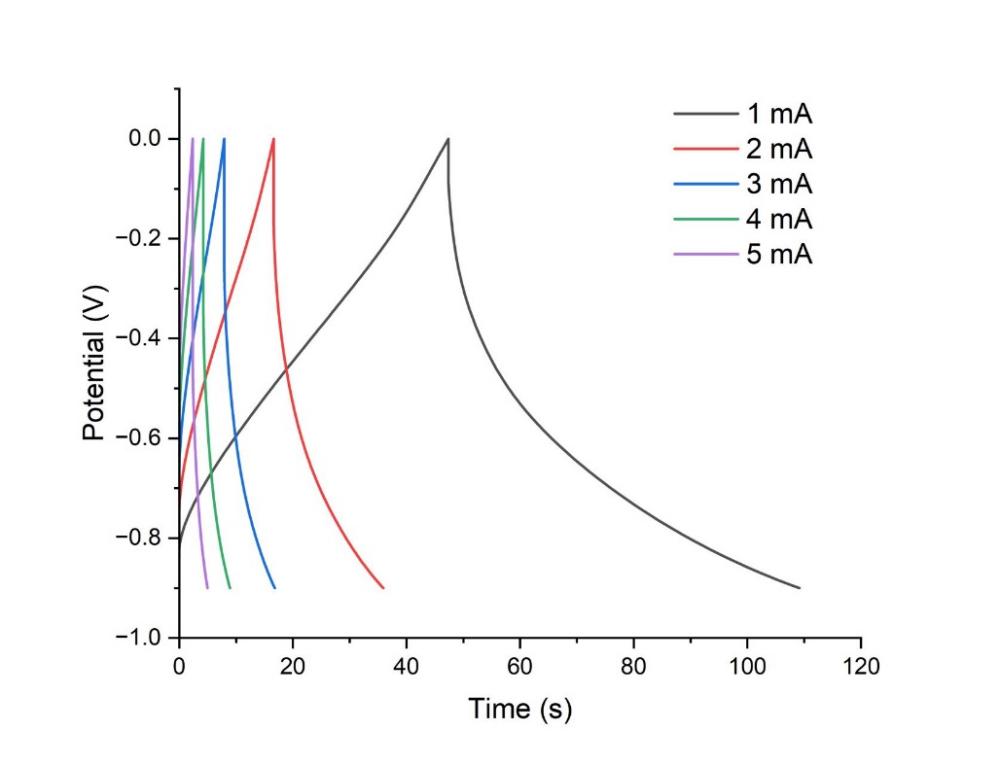
Figure 2: GCD curves of anode at different currents from 1 to 5 mA in the potential window of -1–0 V.
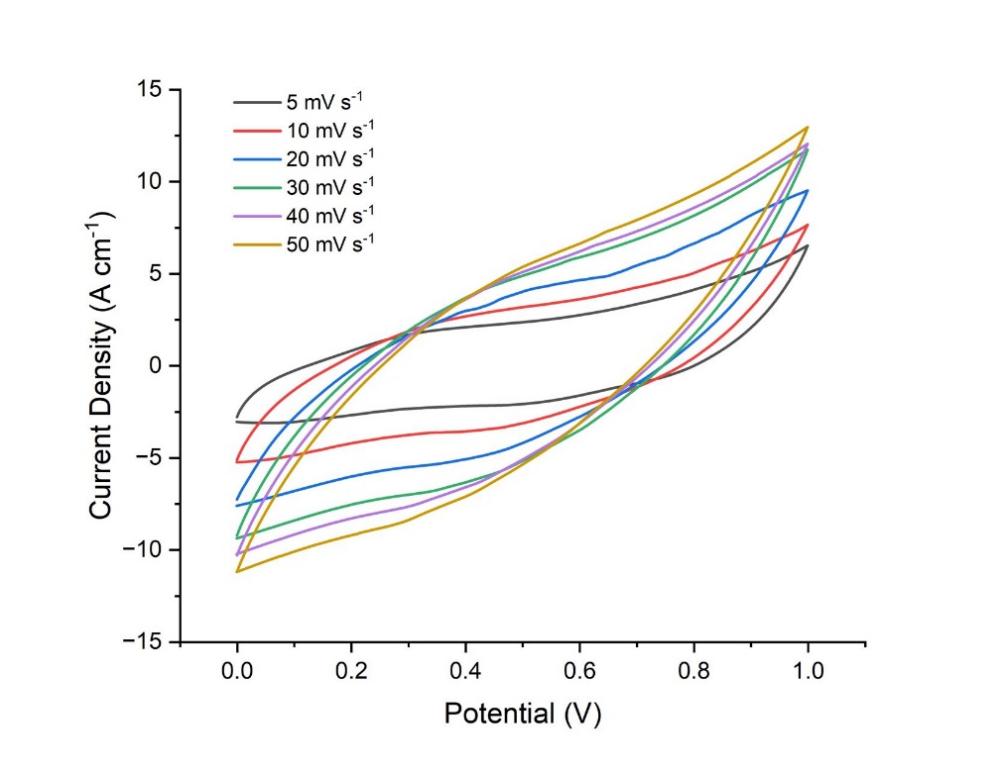
Figure 3: CV curves of cathode at different scan rates ranging from 5 mV s-1 to 50 mV s-1.
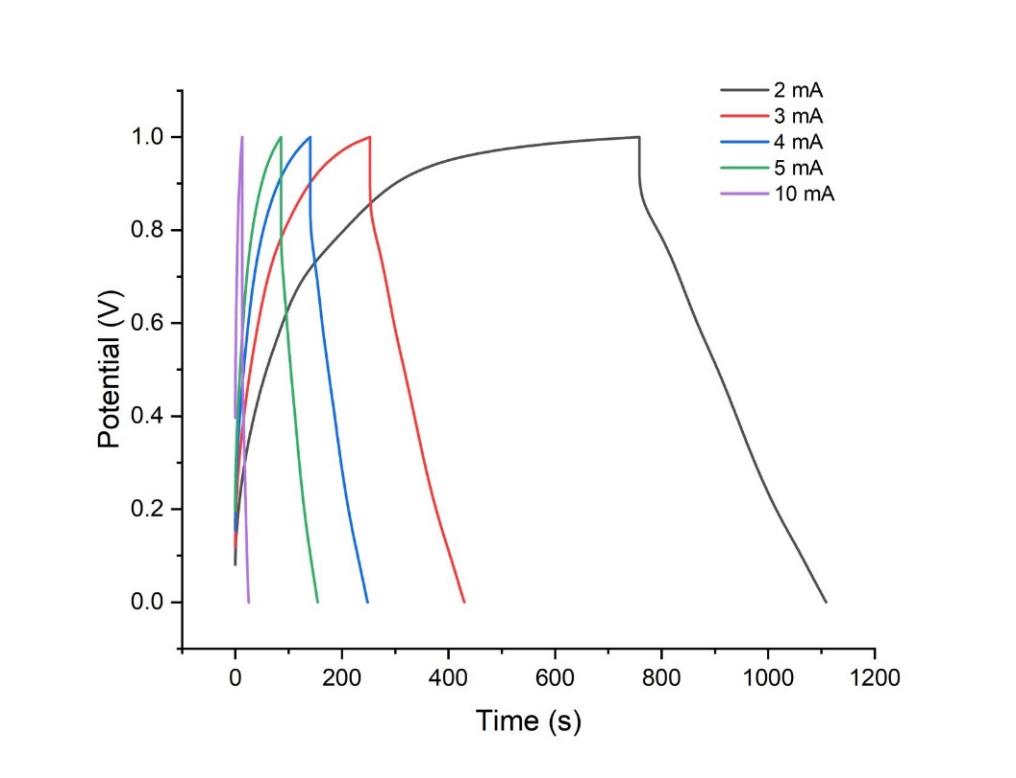
Figure 4: GCD curves of cathode at different currents from 2 to 10 mA in the potential window of 0–1 V.
Figure 1 depicts the cyclic voltammetry (CV) curve of the pseudo-capacitor's anode. The graph is recorded at varying scan speeds ranging from 5 to 50 mV per second, with the potential window increasing from -8 mV to 0 mV. The current rises sharply with the increase of scan rate, demonstrating a characteristic of the electrical double-layer capacitor, as the current curve approaches a rectangular shape with the current's escalation. Figure 3 demonstrates cyclic voltammetry curve of the pseudo-capacitor's cathode, which is definitely different from the anode's. The cathode current under the identical potential window grew significantly more than that of the anode, demonstrating the characteristics of the pseudo-capacitor. Figure 3 illustrates that the cathode's current density exceeded 12 A*cm-1, signifying its exceptional power.
Galvanostatic charge-discharge (GCD) profiles for the anode and cathode were performed at varying current densities of 1 to 5 mA for the anode and 2, 3, 4, 5 and 10 mA for the cathode, respectively. The GCD curves for the anode in Figure 2 exhibit a somewhat symmetric triangular shape, demonstrating its exceptional coulombic efficiency [21]. Figure 2 illustrates that the symmetry of the triangular shapes of the GCD curves, specifically their reversibility, remained constant. However, regarding the cathode in Figure 4, the morphology of the GCD curves becomes asymmetric. The cathode exhibits the characteristics of a pseudo-capacitor [22]. The duration required to charge the cathode to 1V is significantly prolonged for a current of merely 2 mA. Consequently, the cathode necessitates an increased current density. The cathode has promising application potential.
4. Conclusion
To sum up, an all-solid-state asymmetric hybrid supercapacitor is fabricated using MnO2/PEDOT: PSS nanostructured composite as the cathode and the Mxene composite as the anode. In the future, supercapacitors will play a significant role in every aspect of society [23]. The hybrid supercapacitor exhibits excellent coulombic efficiency and exceptional power. When strong current density of 5 mA is added to the supercapacitor, the charging time will be significantly reduced to less than 200 seconds, indicating promising potential for application. Furthermore, the materials involved in the supercapacitor, namely chitosan and MnO2, are relatively easy to produce or obtain from nature, leading to the possibility of being applied in extensive amounts.



